A Waltron Process Study: Fouling Effect of Film Forming Amine on a Sodium Analyzer
Background and Purpose
Film-forming amines (FFAs) are increasingly used in power plants and industrial steam systems as part of advanced water treatment strategies to prevent corrosion by creating a hydrophobic barrier on metal surfaces. While FFAs offer significant benefits in terms of corrosion control and system longevity, their presence in water and steam circuits can interfere with the accuracy of certain online analytical instruments. One area of concern is their impact on Sodium analyzers, which are critical for detecting Sodium contamination in high-purity water systems. Sodium analyzers operate with high sensitivity and rely on ion-selective electrodes or other electrochemical detection methods, making them susceptible to interference from organic compounds.
In this study, we investigated the effects of three different commercial FFA products on the performance of a standard Sodium analyzer. Compared to baseline conditions, the analyzer exhibited significant deviations in performance upon exposure to FFAs. Notably, there was a pronounced drift in Sodium concentration readings and a marked deterioration in response time. The stabilization of simulated break-through events required considerably longer periods than under FFA-free conditions, suggesting chemical fouling or interference at the electrode surface. In addition, daily calibration results revealed a progressive decline in response times that drive measurement accuracy and calibration strength, ultimately resulting in calibration failures over time.
Methods
For the initial testing cycle, Waltron Sodium Analyzer 9033X was selected as the primary measurement instrument. A test stand was constructed, consisting of a 100-liter deionized (DI) water tank with an automatic refill system, connected to a fully adjustable peristaltic pump. In parallel, a container of sample was also connected to an adjustable peristaltic pump. Three* different samples were prepared:
- 1 ppm Sodium standard solution (this is the control)
- 1 ppm Sodium standard solution mixed with 3 ppm of total FFA using FFA product A
- 1 ppm Sodium standard solution mixed with 3 ppm of total FFA using product B
A third FFA product was tested along the same lines, but the data were erratic and unusable. As a result, we have omitted this product from this study. A future analysis of the third FFA product will be carried out separately to assess the cause of the irregularities.
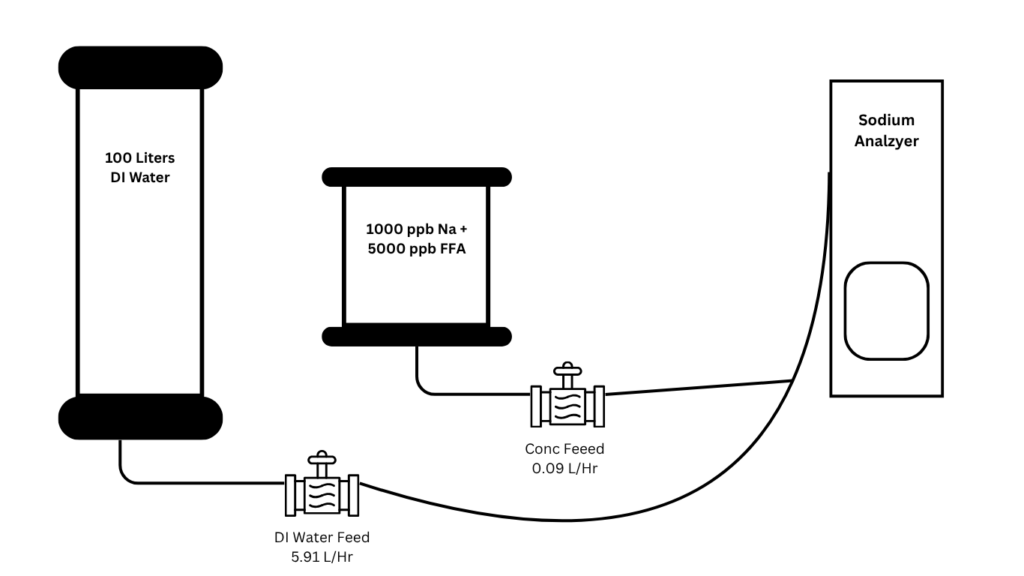
Fig. 1 Experiment Setup
The outputs of both pumps were combined into a single stream and directed to the analyzer, thus a continuous flow of 15ppb Sodium standard and 45ppb FFA standard were fed into the analyzer. This setup allowed for real-time monitoring of analyte response under consistent flow conditions. The data were collected at 10-minute intervals to capture both baseline stability and any transient responses. The analyzer was operated continuously over several consecutive days at a baseline Sodium concentration of 15 ppb (15 µg/L). Three times a day, a spike of 50 ppb Sodium was introduced to simulate a break-through event, and the results were recorded. This assesses the sensitivity and dynamic response of the analyzer. To ensure the repeatability and accuracy of the experiment, three trials were run for each sample. Prior to each new set of trials, both the measuring and reference electrodes were thoroughly cleaned with deionized (DI) water to remove any residual contaminants or analyte carryover. The analyzer was then recalibrated using certified standard solutions to ensure measurement accuracy and to correct for any potential drift in sensor response over time. This approach minimized statistical variability and provided a robust dataset for performance comparison.
Results and Discussion
For the baseline test with sample containing only Sodium standard without any addition of FFA, the analyzer produced a stable and consistent signal over time. Grab samples of 50ppb Sodium standard were also precise and accurate. The measurement exhibited minimal noise and no significant drift, confirming the reliability of the system under controlled, interference-free conditions.
In contrast, samples containing 45ppb FFA products demonstrated noticeable deviations from this baseline behavior. These samples exhibited a pronounced drift in both the magnitude of the measurement signal and the analyzer’s response time. For product A, a notable upward drift in measured Sodium concentration was observed over the course of the experiment (Fig. 2). Within just six days of continuous operation, the analyzer recorded an increase in Sodium levels from the baseline value of 15 ppb to approximately 25 ppb. This represents a significant deviation that cannot be attributed to normal instrumental fluctuations.
Carefully inspecting the analyzer, a substantial accumulation of microbubbles was found surrounding both the measuring and reference electrodes. These bubbles had adhered to and almost completely enveloped the electrode surfaces. Since the exposed surface area of the electrodes plays a critical role in the electrochemical sensing process— affecting both the precision and accuracy of the measurements—the presence of these bubbles likely contributed to the erratic sensor behavior and signal drift. The formation of these gas bubbles is suspected to be linked to the presence of organic compounds in the film-forming amine (FFA) formulation used in product A. These compounds may decompose or react under operating conditions, releasing gases or altering the local surface chemistry near the electrodes. Such interactions can:
- introduce measurement artifacts,
- degrade sensor performance, and/or
- compromise the reliability of long-term monitoring in chemically treated systems.
Upon observing the presence of gas bubbles on the electrodes, a manual intervention was performed in which the electrodes were gently agitated to dislodge and clear out the bubbles from their surfaces. Despite this effort, the upward drift in Sodium concentration measurements persisted. Specifically, the analyzer continued to register a Sodium concentration of 20 ppb, compared to the expected 15 ppb after the removal of bubbles. This indicates that the observed signal deviation was not solely due to the physical presence of bubbles but also likely involved chemical interactions between the first sample formulation and the electrode surfaces. It is likely that components of product A, particularly its organic film-forming compound, adsorbed onto the electrodes or formed a thin film, thereby altering the electrochemical interface.
Such surface fouling could interfere with ion exchange, reduce electrode sensitivity, and contribute to the continued measurement drift even after bubble removal. This observation further supports the hypothesis that product A’s chemical composition inherently disrupts accurate Sodium detection in online monitoring systems. For the other FFAs samples, few microbubbles were found forming around the electrodes, yet the Sodium concentration still increased, only at different rate. (Fig.2).
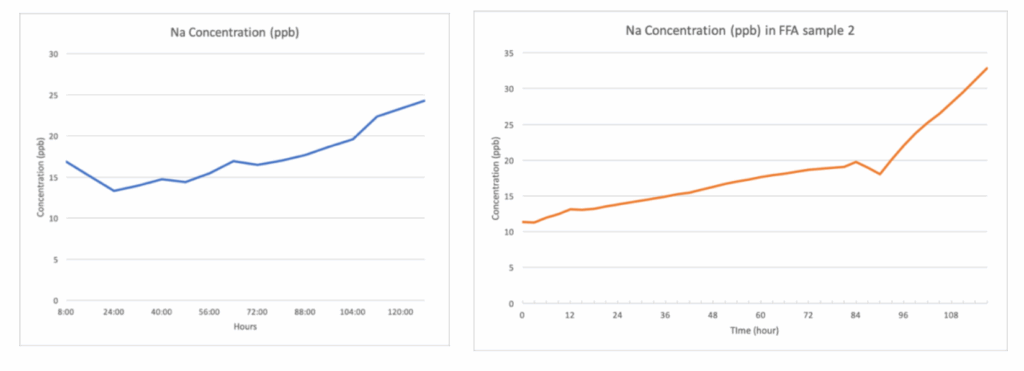
Fig. 2 Na concentration result on sample with FFAs A and B, respectively.
When the analyzer was spiked with a grab sample of 50ppb Sodium standard, a downward drift of data was observed (Fig. 3). This was contrary to expectations, as the continuous monitoring data from the main stream had shown an upward drift, a corresponding increase in the measured Sodium concentration following the 50 ppb spike was anticipated. Thus, further investigation was carried out to the explain this abnormality. For the Waltron 9033X Sodium Analyzer, the grab sample was set to run for 15 minutes to allow sufficient time for the measurement to stabilize before recording the final result.
Under normal operating conditions, data stabilization is achieved within the first 5 minutes, as confirmed during baseline tests. However, during this experiment, the Sodium concentration measurements continued to increase at a very slow rate, even towards the final minutes of the 15-minute cycle. This behavior indicated that the data had not yet stabilized, suggesting a significant degradation in the analyzer’s response time. The slow stabilization implies that the instrument was struggling to reach equilibrium with the Sodium concentration of the sample within the acceptable amount of time. This degraded response time could be attributed to the fouling or chemical interference at the electrode surfaces, possibly linked to the film- forming amine present in the system. Such interference would slow down ion exchange kinetics at the electrodes, delaying the analyzer’s ability to accurately and promptly reflect changes in Sodium concentration.
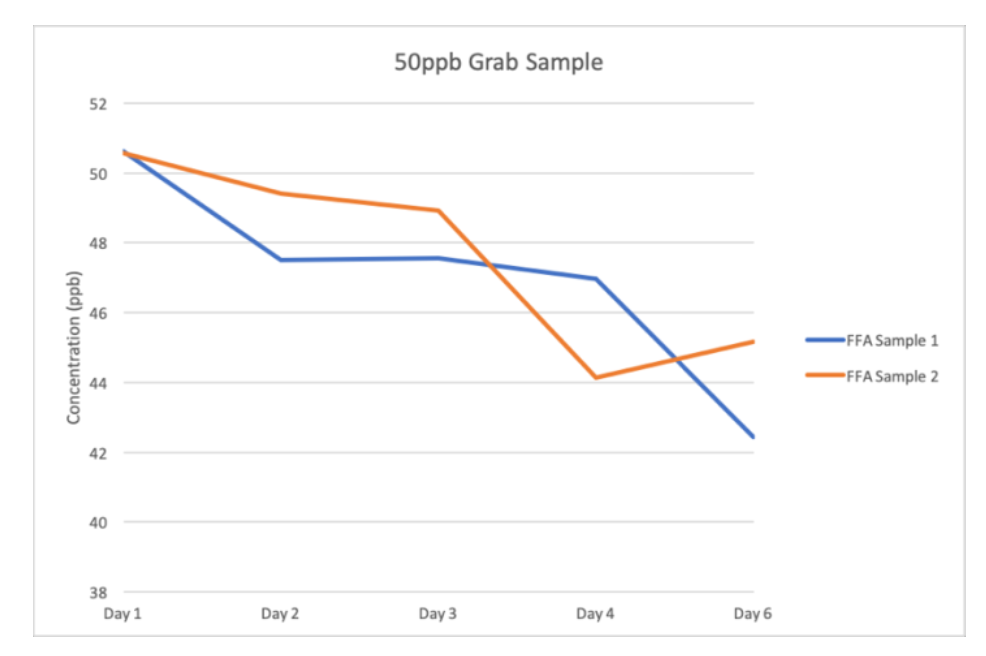
Fig. 3 50ppb Grab sample results on a Sodium analyzer that has been continuously feeding with FFA type A and B for 6 consecutive days.
Due to this phenomenon, an additional experiment was conducted to evaluate how long it would take for the analyzer to reach a “Calibration Fail” status under repeated daily calibration. While the analyzer was continuously fed with 15ppb of Na standard and 45ppb of FFA sample B, calibration was carried out each day using Sodium standard solutions at concentrations of 100 ppb and 1000 ppb, both of which were free of FFAs, and no cleaning on electrodes was done during this period of time.
Each standard was introduced to the analyzer for a duration of 15 minutes to allow for measurement stabilization. Under normal conditions, a successful calibration is indicated by potential millivolt (mV) readings of approximately 208 mV for the 100 ppb standard and 149 mV for the 1000 ppb standard, yielding a calibration strength value of 97-100. During the initial three days of the experiment, the calibration strength remained within acceptable limits.
However, a notable upward trend was observed in the potential mV readings for both standards (Fig. 4), indicating that the analyzer was registering lower-than-actual Sodium concentrations—since higher mV readings correspond to lower Sodium levels. This trend supports the hypothesis that FFAs had formed a coating on the electrode surfaces, impairing their ability to accurately detect Sodium ions and resulting in the analyzer setting new calibration values at higher mV readings for each day. Thus, the calibration strength remained within tolerance even as the sensors showed signs of fouling. This “false positive” calibration will create a situation where the analyzer will record lower than actual Sodium concentration sample in an adverse event.
By the third day, the analyzer failed calibration, reporting a calibration strength of only 49. The potential reading on 1000 ppb standard had not reached stability and was still decreasing in mV readings at the end of the 15-minute cycle, while the 100 ppb standard also exhibited an elevated mV reading. As shown in Fig. 4, the gap between the 100 ppb and 1000 ppb standards began to narrow after the third day.
By the sixth day, the calibration strength had dropped further to 41, with only a 24 mV difference between the two standards. This indicates that FFAs had progressively saturated the electrode surfaces to the point that the response time for the analyzer to reach accurate potential for 1000ppb has increased significantly, and not within the acceptable range. Instruments like the Waltron 9033X Sodium Analyzer that use an automatic, two-point calibration process with defined period of time and frequency will gradually calibrate themselves to lower than actual values of standards and eventually reach “calibration failed”.
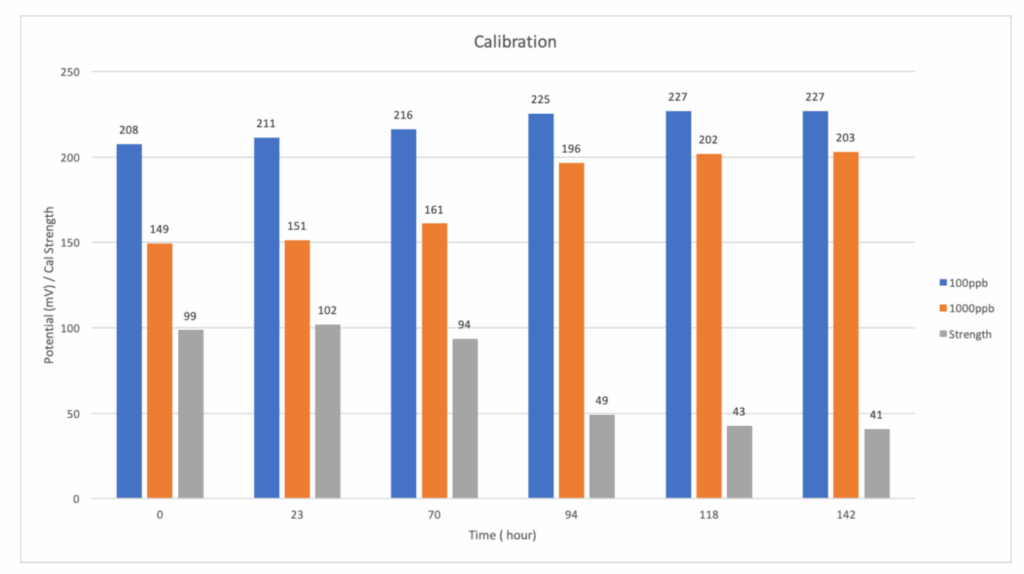
Fig. 4 Potential in mV for 100ppb and 1000ppb in 6 consecutive days. Calibration Strength of corresponded 100ppb and 1000ppb data
Further investigation was carried out to observe the response time of the analyzer when it is spiked with 100ppb. Once each day the analyzer was provided a sample of 100 ppb Sodium standard for exactly 10 minutes, and the electrode responses for these Sodium concentration spikes were then recorded. Under normal condition, the sensor shows a sharp drop in potential from sample with 15ppb to 100ppb in the first 2 minutes and it reaches stability within 5 minutes. After exposing to FFAs for more than 7 days, some fouling effects started to become apparent. It takes much longer for the sensor to reach stability from 15ppb to 100ppb sample, and it eventually failed to reach the appropriate potential mV for 100ppb Sodium standard after the 11th day. A comparison of the first week’s average electrode response versus those from days 11-13 can be seen in Fig. 5. This created a situation where the analyzer appeared to be functioning normally but was no longer capable of responding to adverse events such as a condenser leak. These highly dampened responses will follow trends over time but are incapable of rapidly reacting to transient spikes or changes in values.
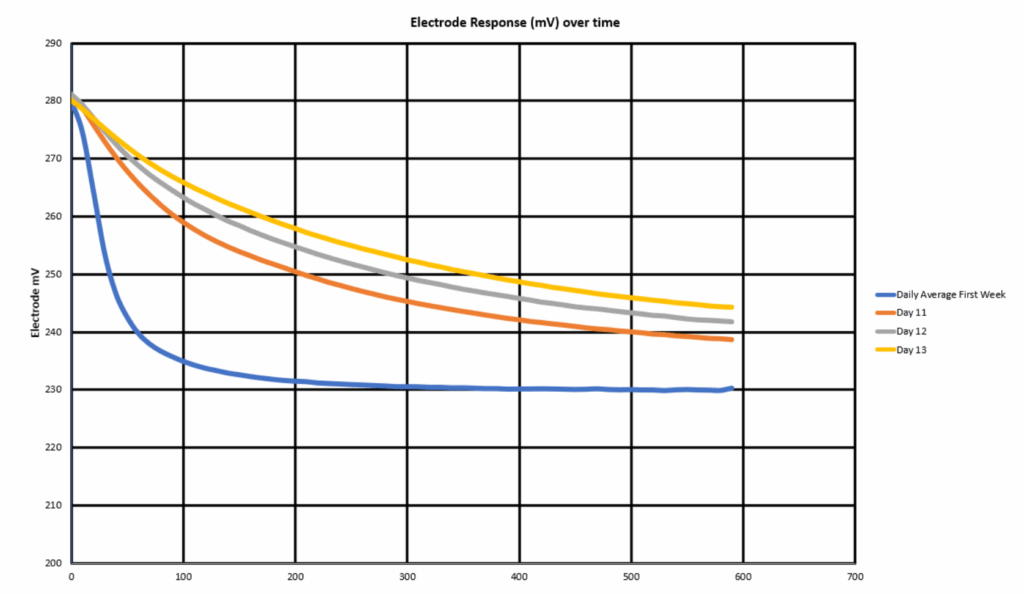
Fig. 5 Electrode response time to 100ppb after FFAs exposure
Cleaning Process
A process to remove most FFA fouling on the Sodium electrodes was identified. The normal electrode regeneration process, typically soaking it in a solution with Sodium Fluoride made the fouling situation worse. A combination process of mechanical and chemical cleaning of the electrode with acetic acid, followed by normal electrode regeneration, appeared to restore most of the original functionality. However, such handling of delicate glass electrodes is not recommended in the field. Because fouling occurred so rapidly under our testing conditions, mechanical cleaning about every week would bring significant risk of electrode damage, not to mention down-time of the analyzer and added labor to conduct this maintenance.
Conclusion
This study demonstrates that film-forming amines (FFAs), while beneficial for corrosion control in steam and water systems, can significantly compromise the performance of online Sodium analyzers such as the Waltron 9033X. Exposure to three commercial FFA products at total FFA concentration of 45ppb led to notable drift in Sodium concentration readings, degradation in response time, and progressive calibration failure. The presence of FFAs promotes chemical fouling at the electrode surface, resulting in measurement inaccuracies in Sodium concentration. Following one week of continuous exposure, the analyzer was no longer able to achieve stable readings for a 1000 ppb Sodium standard within the acceptable 15-minute stabilization period, indicating substantial degradation in measurement reliability and responsiveness. Calibration data revealed that despite daily recalibration efforts, sensor performance deteriorated over time, ultimately resulting in “Calibration Failed”.
This persistent significant drift and degraded response time are most likely caused by the adsorption of organic FFA compounds onto the electrode surfaces. The surface fouling alters the electrochemical interface, impeding ion exchange and reducing electrode sensitivity. While FFAs offer valuable corrosion protection, their compatibility with sensitive online monitoring instruments must be carefully managed. Future work should explore mitigation strategies such as enhanced electrode cleaning protocols, sensor material innovation, or alternative monitoring technologies better suited for environments where FFAs are present.
For a PDF of the Process Study – Click on the Botton Below
Ready to take your next step?
Questions? Need Support? A Quote? Get in touch with a Waltron Representative.
